 
It also helps identify conserved regions in proteins that may have distant evolutionary relationship.It helps compare two protein chains that have adopted different conformational states, e.g., due to post-translational modifications such as phosphorylation or interaction with other proteins/ligands.When superposition by rigid alignment alone does not yield meaningful results, introducing flexibility to structural alignment becomes useful for two main reasons: In a flexible structure alignment relative mobility between domains or subdomains in each structure is accommodated. Rigid body alignments are well-suited for identification of structural equivalences between proteins that are closely evolutionarily related and thus have similar shapes. 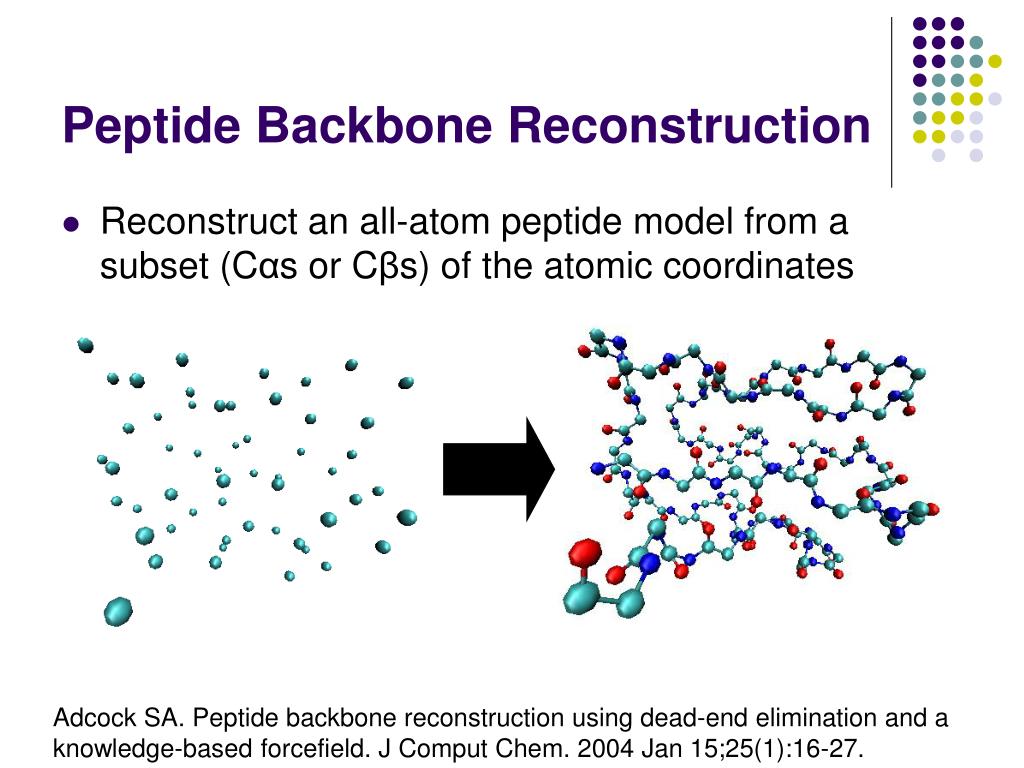
In the resulting superposition, only the overall shapes of the structures are aligned. In a rigid body alignment, the relative orientations and positions of atoms within each structure remain fixed during the alignment process. In contrast to multiple structure alignment (reviewed in Ma and Wang, 2014) that provides a global solution for three or more structures.ĭifferent types of structural alignments and their rationales are described below. In the case of pairwise alignment, structures are always compared in pairs. This tool presents options for pairwise structure alignment of proteins. Structure alignment requires no prior knowledge of equivalent pairs of residues, does not rely on the sequence alignment, and the type of residues is ignored when the correspondence is established. Structure alignment attempts to establish residue-residue correspondence between two or more macromolecular structures based on the optimal superposition of their shape and three-dimensional conformation. References Introduction What is Structure Alignment?.Diversity, Equity, Inclusion, and Access.Citation, Usage, Privacy Policies, Logo. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
